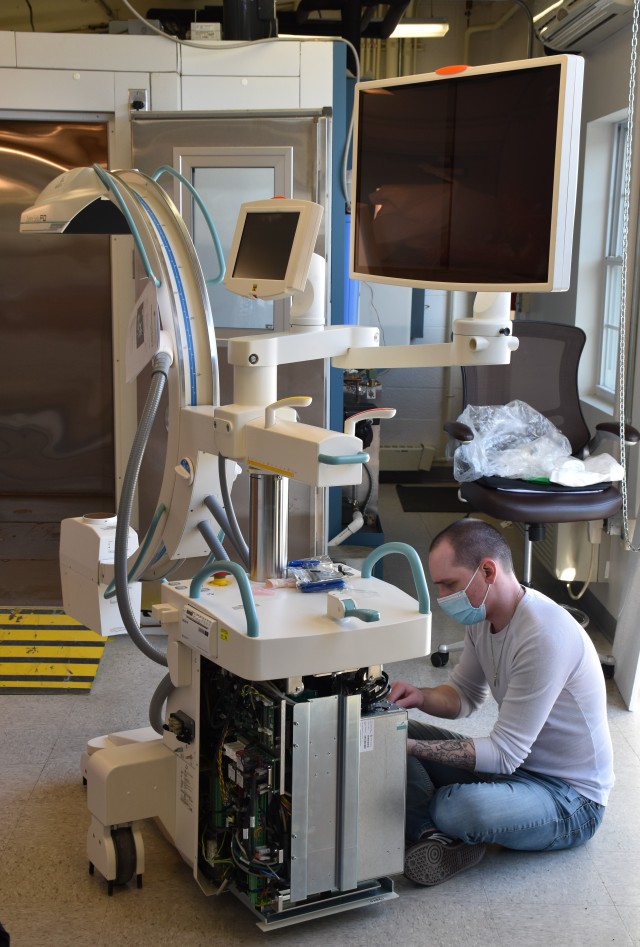
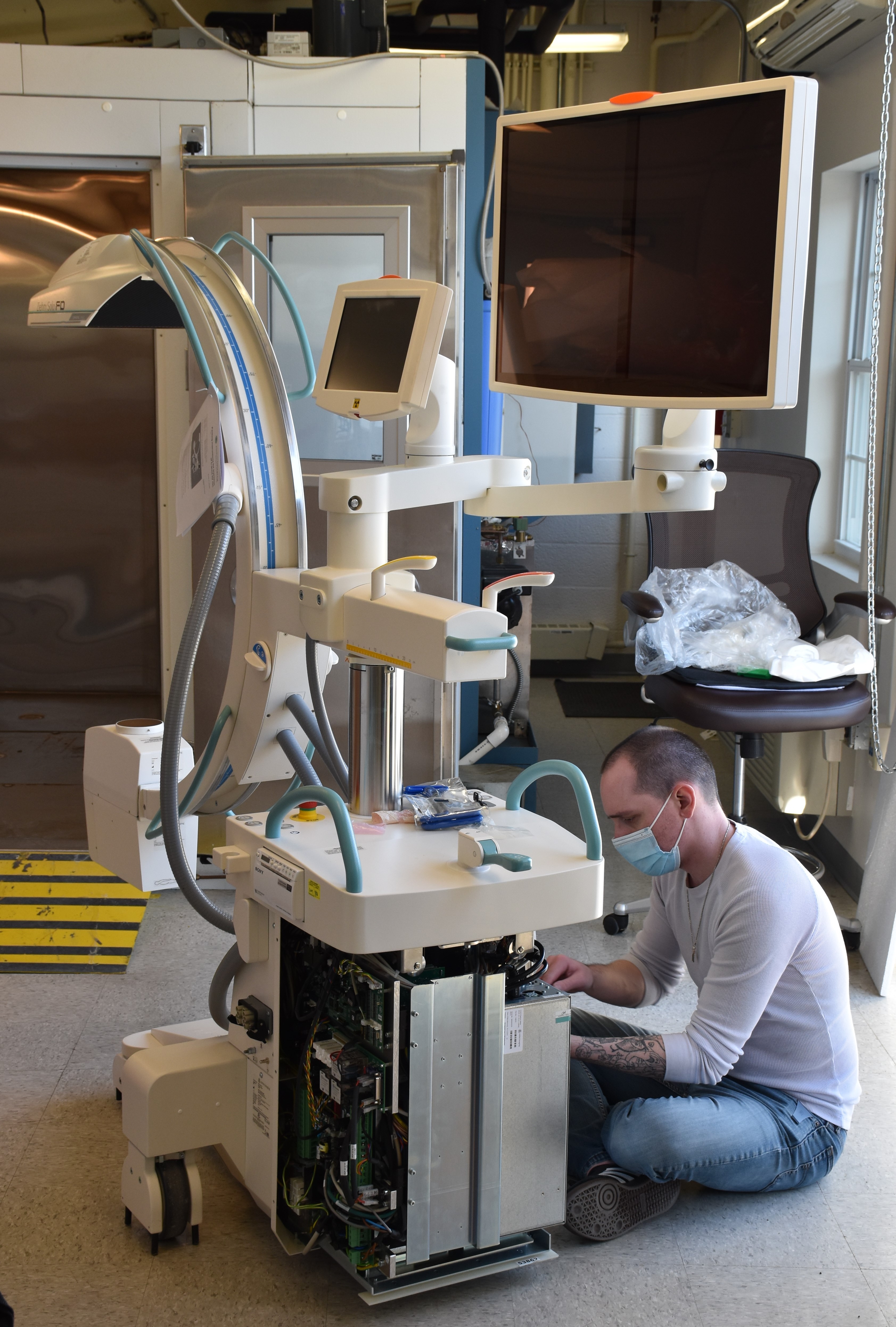
The U.S. Army Medical Materiel Development Activity recently received the state-of-the-art “C-Arm” high-capacity radiographic and fluoroscopic unit, which is a mobile, self-contained X-ray machine that uses non-invasive real-time fluoroscopic imaging in the operating room during examination, diagnosis and surgery. This innovative system is used primarily to locate and treat vascular injuries, such as uncontrolled bleeding, during surgery, as well as in the removal of foreign bodies and to triage patients. The successful implementation of the C-Arm unit at military treatment facilities will help to reduce the fatality rate of Warfighters who have suffered vascular injuries.
Wendy Hagan, program manager for USAMMDA’s Warfighter Deployed Medical Systems Project Management Office, currently oversees the cost, schedule and performance of the C-Arm effort. She is responsible for leading the C-Arm Working Group, which includes subject matter experts , clinicians, contractors and other personnel who work together to facilitate modernization of the device. In her role, Hagan facilitates the formation and evaluation of essential characteristics to ensure the C-Arm is tailored to meet the needs of the Warfighter, while safeguarding its sustainability in deployed scenarios through a comprehensive test and evaluation process.
“USAMMDA’s C-Arm machine has undergone environmental testing at the U.S. Army Medical Research and Development Command’s Test Branch at Fort Detrick, Maryland, to evaluate the durability of the unit and its components in hostile environments,” Hagan explained. “Upon completion of the environmental testing requirements, the C-Arm was re-packaged and transported to Camp Bullis, Texas, to complete operational testing before initial procurement by military operational units.”
Hagan and other WDMS PMO personnel are currently in San Antonio to oversee user testing of the C-Arm unit, and she said the process is going very well thus far. She anticipates testing will conclude by November 20, and her team does not foresee any issues that would prevent the C-Arm from passing all of the testing requirements.
“This is the first C-Arm unit that we’ve received,” said Hagan. “After operational and user testing is successfully completed in San Antonio, we will begin procuring additional units for our military forces, with the objective of acquiring 42 C-Arm units. We also will have an option for an additional 54 units in future procurements.”
“We are very excited about the vast potential of the C-Arm’s ability to save lives,” she continued. “During our current war on terrorism, vascular injury is five times higher than in previous wars, resulting in 20 percent of battlefield injuries. This piece of medical equipment will help to reduce morbidity and mortality due to these injuries, which will also help to improve Soldier readiness.”
USAMMDA is a subordinate command of the U.S. Army Medical Research and Development Command, under the Army Futures Command. As the premier developer of world-class military medical capabilities, USAMMDA is responsible for developing and delivering critical products designed to protect and preserve the lives of Warfighters across the globe. These products include drugs, vaccines, biologics, devices and medical support equipment intended to maximize survival of casualties on the battlefield.
U.S. Army Medical Research and Development Command


Social Sharing