Within the critical field of burn treatment and skin repair, the U.S. Army Medical Materiel Development Activity's Tissue Injury and Regenerative Medicine Project Management Office remains a leading force in discovering effective medical solutions for our nation's Warfighters to restore form, function and appearance following catastrophic injuries sustained in service. As a subordinate command of the U.S. Army Medical Research and Materiel Command, the entire USAMMDA team works tirelessly to fulfill its mission of protecting and preserving the lives of U.S. Service Members, and civilians, worldwide.
Dr. Melanie Eacho serves as the product manager and Integrated Product Team chair for the TIRM PMO's Burn Treatment and Skin Repair portfolio, which includes three major focus areas: 1) Burn Conversion Prevention Products, 2) Burn Wound Scarring Treatment and 3) Functional Skin Regeneration. In her role, Eacho guides the efforts of both TIRM PMO team members and the commercial partners who are contracted to develop the important medical products requested by the Department of Defense.
"Throughout the three Burn Treatment and Skin Repair focus areas, there are many projects that we follow on a daily basis," said Eacho. "These may fall under a 'tech watch' advisory, where we closely monitor the progress of certain products, or they are under contract within our oversight."
With the sharp rise of incidents involving improvised explosive devices in military conflicts over the past decade, a greater number of Warfighters now suffer catastrophic burn injuries than perhaps ever before. In light of this, the TIRM PMO has been tasked to seek out effective solutions for burn wounds that may be used at various points along the treatment spectrum, from point of injury through to aftercare.
As Eacho explains, however, many efforts progress through "small steps," so the team often focuses on "incremental approaches that may lead eventually to the product of interest." This remains the case for two recent wound gel products under her charge, Catasyn and KeraStat(R) Gel, which were funded in 2013 through the DOD's Small Business Innovation Research program, falling under the Burn Wound Scarring Treatment focus area.
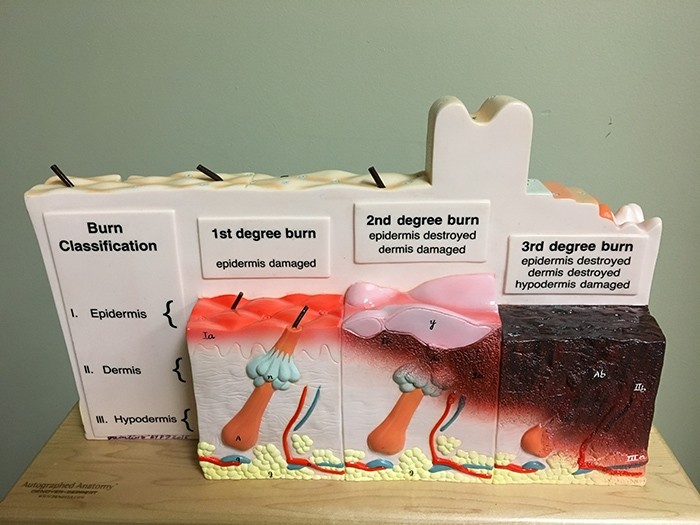
"Stemming from a topic titled, 'Development of Technologies that Control Scar Contracture after Burn Injuries,' Catasyn and KeraStat(R) Gel made it through SBIR Phase 2," said Eacho. "Both companies developed a hydrogel with a drug/protein component to address burn injuries, up to second-degree level, and they each have obtained U.S. Food and Drug Administration 510(k) clearance, which allows these products to be marketed for their intended use."
Both products provide a moist wound environment, which is supportive to wound healing, and both are available currently by prescription, although an over-the-counter version of Catasyn is also available. Further, both prescription-level products are indicated for the management of a number of partial-thickness skin wounds including ulcers, surgical wounds and burns, up to second-degree.
"Although it appears our commercial partner may not be moving forward with its Catasyn gel," said Eacho, "KeraNetics [also commercial partner] is continuing on with a clinical study for its KeraStat(R) Gel, to show reduction both in time-to-wound-closure and the prevention of scarring."
With a long-range goal of developing an effective burn treatment product for the type of severe injuries caused by catastrophic explosives and serious fires, Eacho explains that "you have to start somewhere," and these introductory wound gels appear to serve well as the impetus for later research.
"As for the KeraStat(R) Gel, we're currently looking at its effectiveness with partial-thickness, second-degree, burns," she said. "The U.S. Army Institute of Surgical Research [a subordinate command of USAMRMC] has been working successfully with KeraNetics, and we are hoping to fine-tune the product to suit our needs for military use, both in the field and at military treatment facilities."
While most products developed by USAMMDA are intended initially to fulfill a military requirement, the common end-goal is also to convey a successful medical product or device to civilian use, so it may be utilized in a much larger capacity worldwide. This also allows greater flexibility for the Army with regard to mass production and distribution, as it would be difficult financially to sustain on its own all of the products and devices necessary to support our military population. Therefore, commercial partnerships remain critical in the advanced development and fielding process.
Along with its successful research in extremity repair, transplantation, prosthetics and sensory restoration, the TIRM PMO continues to make great strides in the field of burn treatment for both the military and civilian population -- and the many recipients of this critical exploration will benefit greatly for years to come. As a result of this valuable work, Eacho and her colleagues certainly help to secure USAMMDA's role as the premier developer of world-class military medical capabilities.

Social Sharing