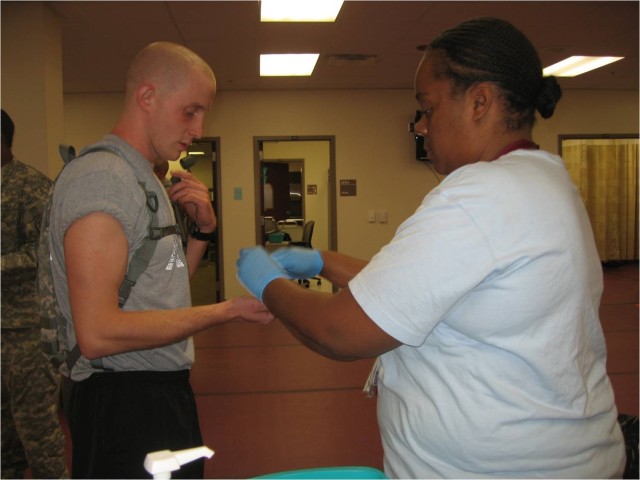
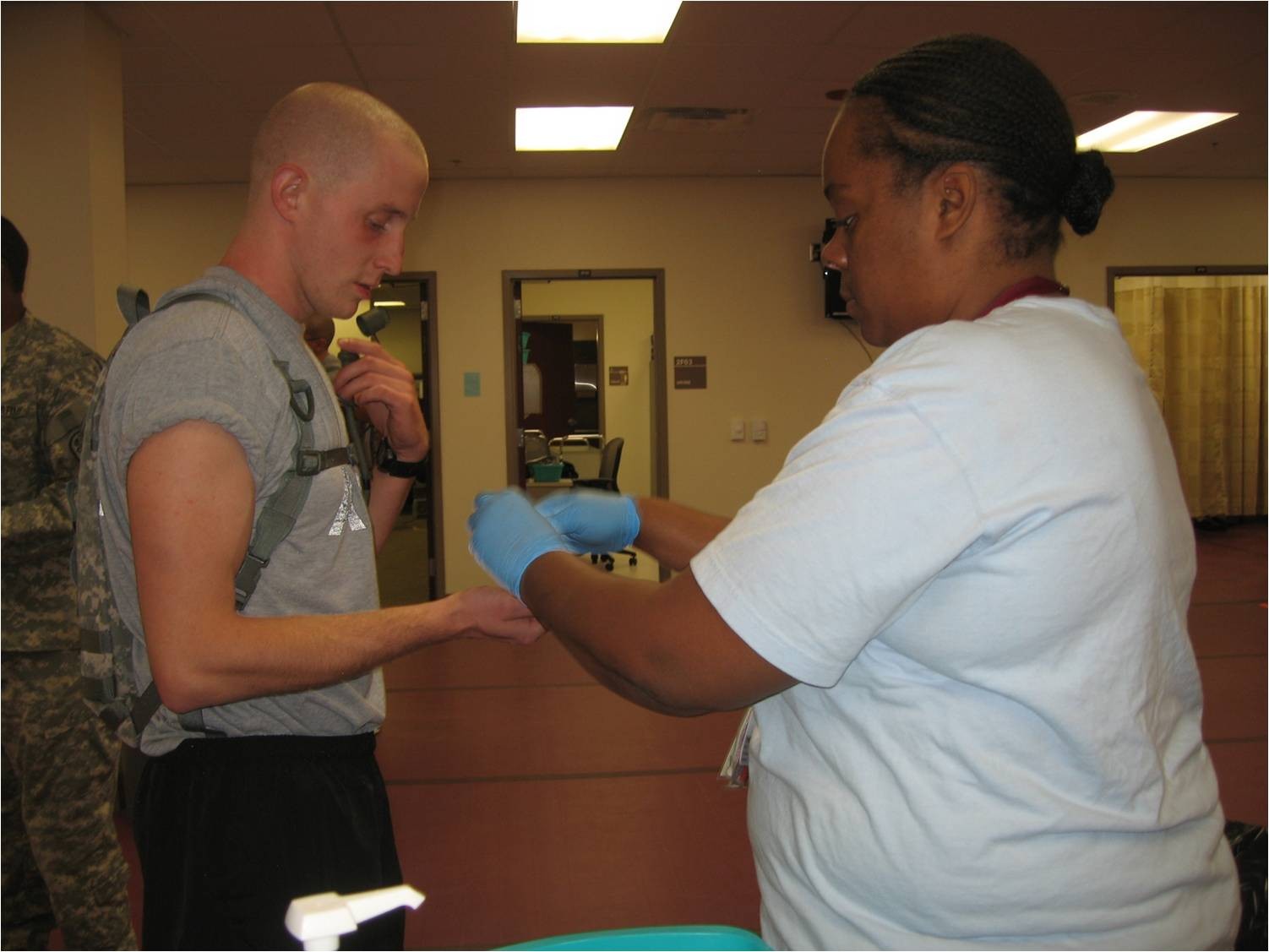
FORT DETRICK, Md., Oct. 31, 2011 -- The U.S. Army Medical Research and Materiel Command announced that the new adenovirus vaccine, which replaces the older version that has been out of production for over a decade, was shipped to basic training sites Oct. 18.
Every vaccine tablet will be delivered to the nine basic training sites, all branches of the U.S. military and the Coast Guard, in this phase of fielding. Immunizations began at Fort Leonard Wood, Mo. and Fort Sill, Okla. Oct 24. Use at the seven other basic training sites will begin within the next week.
"From this day forward, the work that we've done literally reaches into the gut of every military recruit during basic training, because adenovirus vaccine, administered orally, begins its work in the small intestine, where it starts the process of triggering immunity," said Dr. Cliff Snyder, product manager for the adenovirus vaccine.
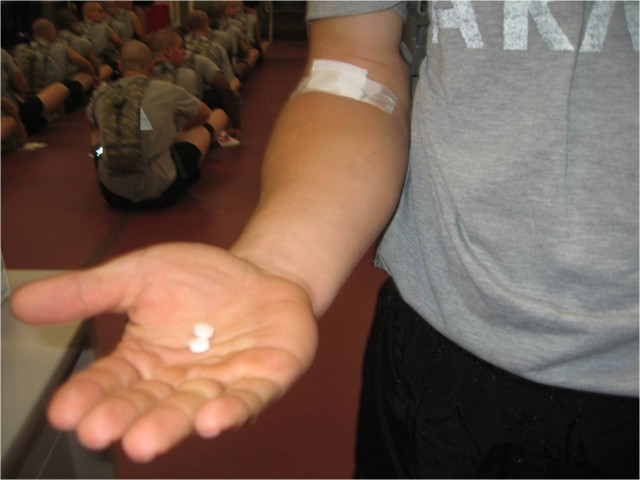
The new adenovirus vaccine offers protection against two strains of the virus, type 4 and type 7, and is administered in tablet form. In clinical trials supported by both the Army and the Navy among other organizations, scientists found the new vaccine provided 99.3 percent protection against febrile respiratory illnesses due to the adenovirus type 4 while stimulating protective levels of antibodies against the adenovirus type 7.
Dr. Lawrence Lightner, USAMRMC project manager for Pharmaceutical Systems said, "[Adenovirus] infections cause approximately 15,000 illnesses per year in basic trainees."
These positive results should come as welcome news to the relatives and friends of the warfighter -- as well as the warfighters themselves -- currently serving in the U.S. military and the Coast Guard.

Social Sharing