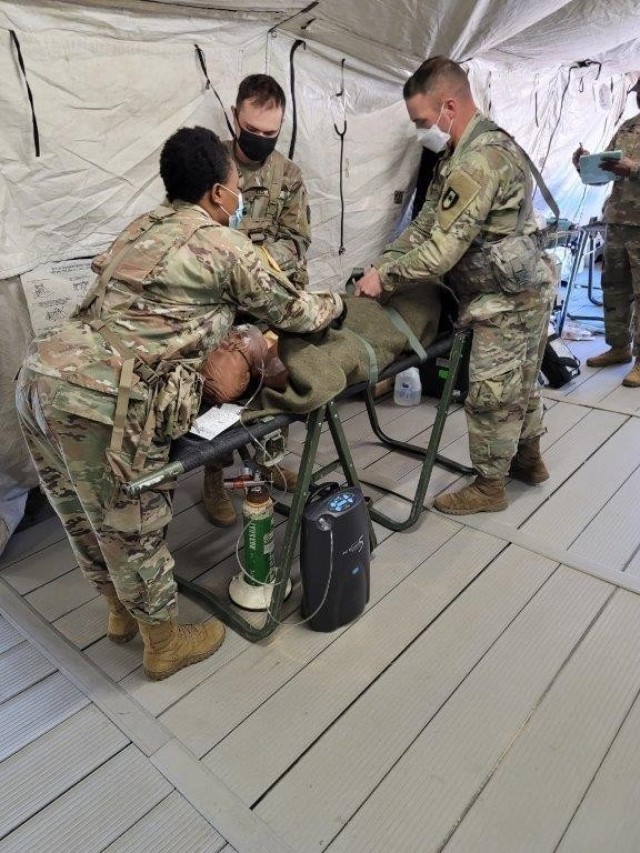
The Army is currently testing an oxygen generator that has a longer shelf life than the one currently in use and will meet the requirement of supplemental oxygen that medics provide to combat casualties.
The Field Oxygen Generator Resource (FOGR) is being considered as a replacement for the Oxygen Generator, Field Portable (OGFP) that is currently used to supply supplemental oxygen to sick and wounded Soldiers in the field. In February, the U.S. Army Medical Department Board (USAMEDDBD) conducted an operational test using Soldiers from the 44th Medical Brigade during a field training exercise to test the effectiveness and suitability of the set up and operation of the FOGR to provide critical care.
According to Archie C. Kinnebrew Jr., lead test officer with USAMEDDBD, the success of the February test event will inform decisions that determine if and when the FOGR is fielded to Army units.
“There is truth in operational testing. Army leadership uses the results from test events to facilitate risk-reduction for product fielding,” Kinnebrew said. “The test articles under consideration will not only be evaluated by the testing community, but will also include input from the intended end-users on the battlefield. These test events ensure that Soldiers have a voice in the acquisition and deployment of new and improved systems.”
Kinnebrew had words of praise for the 44th Medical Brigade Soldiers who put the FOGR to the test through a series of exercises.
“The Soldiers of the 44th Medical Brigade - 36th Medical Company Area Support and the 240th Forward Resuscitative Surgical Team - were enthusiastic while putting the FOGR to use during the test,” Kinnebrew said. “They provided honest feedback, which greatly assisted the test team in capturing the data needed. Their participation was key to the success of this test event and is greatly appreciated. The professionalism and dedication displayed by these Soldiers really made me feel proud.”
Austin S. Langdon, assistant product manager with Warfighter Deployable Medical Systems, U.S. Army Medical Materiel Development Activity at Fort Detrick, Md., said the Army is replacing the OGFP because of sustainability issues.
“The old device was designed to operate 10-12 hours a day and 7 days a week. This is the case for most portable Oxygen Concentrators (POCs) on the market. However, when the device sits on a shelf, maintenance issues arise from lack of use, which are very costly,” Langdon said.
Langdon said that USAMMDA is currently testing two commercial off-the-shelf (COTS) products. One of the devices being tested has the ability to be placed on the shelf for up to three years without any need for maintenance, a bigger advantage over the current model in use.
“Since this is a COTS item, the devices being tested are already in production and can currently be purchased. However, the Army still needs to test it for airworthiness and also MIL-STD-810H testing,” Langdon said.
MIL-STD-810 is a United States Military Standard that emphasizes tailoring an equipment's environmental design and test limits to the conditions that it will experience throughout its service life, and establishing chamber test methods that replicate the effects of environments on the equipment rather than imitating the environments themselves. Although prepared specifically for military applications, the standard is often used for commercial products as well.
The OGFP weighs 12 pounds and was an advanced development item that was specifically designed for the Army’s use. FOGR outweighs the current device by a few pounds, but has less maintenance issues.
Langdon said that the Army is looking to get quantitative data from the testing that will allow for informed decisions on these variants and if they will fit the end user’s needs.
When asked when FOGR will be available to the Army, he added, “These devices are both already in production. If one is selected, it will be fielded to the force later this year.”
Social Sharing