FORT SAM HOUSTON, Texas (Jan. 19, 2012) -- Saving the lives of combat wounded is one of the main objectives at the U.S. Army Institute of Surgical Research at Fort Sam Houston, Texas. Research on prehospital care devices like the tourniquet has proven to save lives in the battlefield wounds to the arms or legs, which was once the most common cause of preventable death.
Now, after almost two years of research on a device to prevent the most common cause of preventable death in the battlefield, the junctional tourniquet is ready for battlefield use.
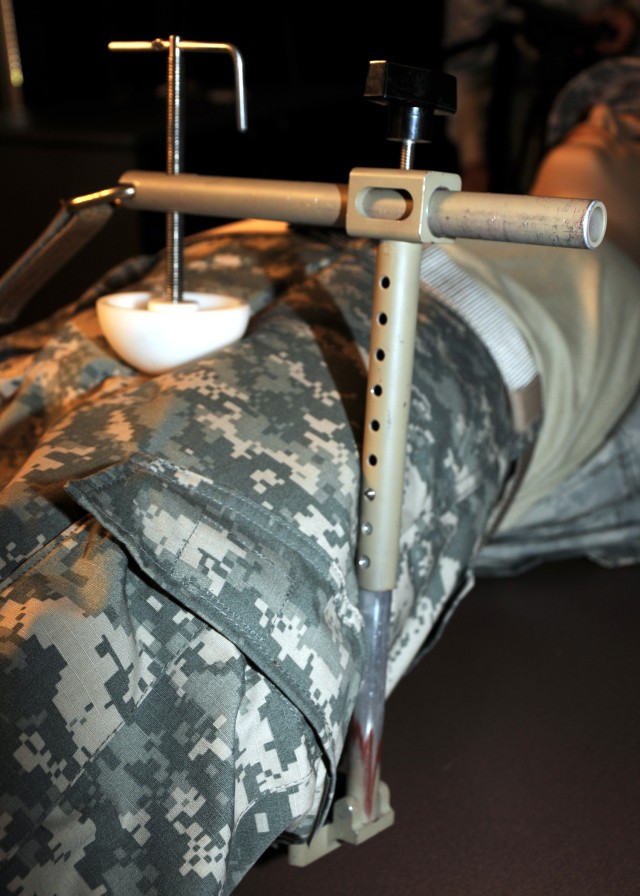
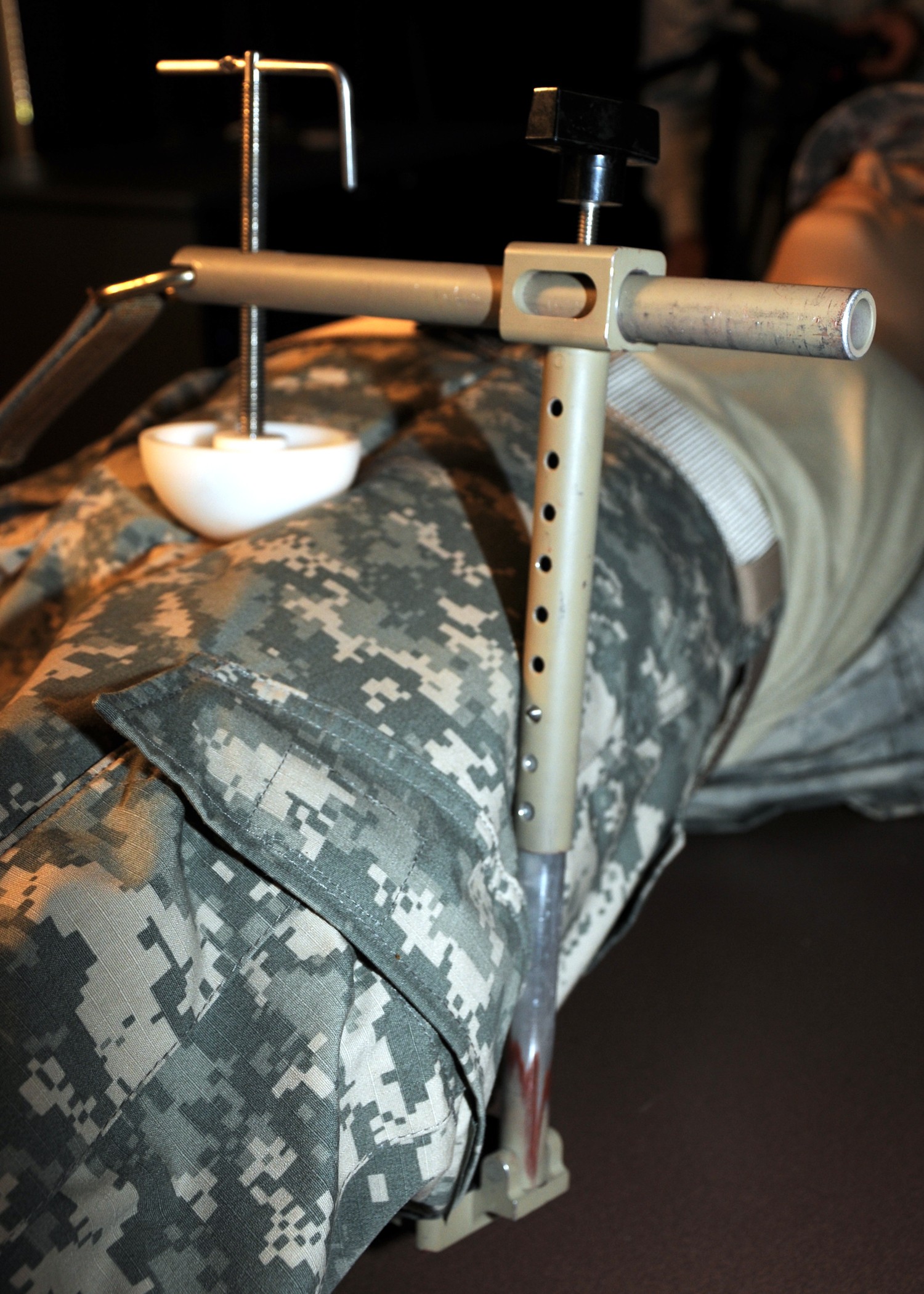
"Exsanguination, or bleeding to death, from the body is now the most common cause of preventable death to wounded warriors on the battlefield," said Dr. John F. Kragh, an orthopedic surgeon and researcher at U.S. Army Institute of Surgical Research, or USAISR. "Groin hemorrhage is the most common type of junctional bleeding where regular tourniquets cannot work."
Research on the effectiveness and type of battlefield truncal tourniquet began in 2009 after the Committee on Tactical Combat Casualty Care made it a research priority. Shortly after this, the Combat Casualty Care Research Program, of the U.S. Army Medical Research and Materiel Command, and USAISR headquarters requested candidate devices for the research.
The Combat Ready Clamp, or CRoC, which has been approved by the U.S. Food and Drug Administration, is manufactured by Combat Medical Systems in Fayetteville, N.C. This clamp was selected from a handful of junctional tourniquet prototypes from those submitted after the request for candidate devices.
"The CRoC is a vice-like tourniquet that can be placed over the groin and lower abdomen," said Kragh. "A pressure ball is screwed down to press on the artery and to stop the bleeding."
The USAISR is committed to optimizing combat casualty care by focusing on providing requirement-driven combat casualty care medical solutions and products for injured Soldiers, from self-aid through definitive care, across the full spectrum of military operations.
Related Links:
U.S. Army Medical Research and Materiel Command




Social Sharing