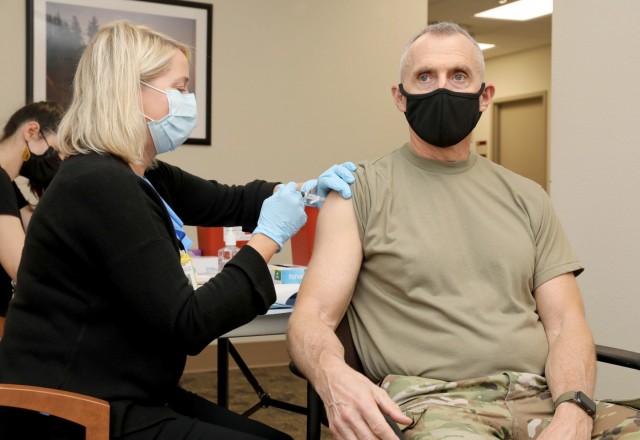
JOINT BASE LEWIS-McCHORD, Wash. — Soldiers and Department of the Army civilians assigned to Regional Health Command-Pacific recently rolled up their sleeves to become part of a four-year-long study to compare the effectiveness of three existing vaccines for preventing influenza infection.
The study, "A Pragmatic Assessment of Influenza Vaccine Effectiveness in the Department of Defense," or PAIVED, is conducted by the Infectious Disease Clinical Research Program at the Uniformed Services University and supported through the Defense Health Program. The study is being conducted at 10 DOD sites, including JBLM.
Susan Chambers, a registered nurse who is the clinical site manager supporting USU’s Infectious Disease Clinical Research Program at Madigan Army Medical Center, said the study focuses on active-duty service members and other beneficiaries.
Despite the high receipt of annual flu vaccination in the military population, there continues to be a significant rate of influenza illness, she said.
“We are comparing three types of [Food and Drug Administration]-approved flu vaccine to see if there is one that performs better,” Chambers said.
The comparison will provide important information on immune response to and effectiveness of the different flu vaccines.
More than 18,000 people are participating in the study, which organizers said will assist in a better understanding of the influenza vaccine, how to better protect participants and loved ones from the flu, and help advance military medicine.
Influenza remains a significant threat to operational readiness in the U.S. armed services, organizers said.
Each of the three vaccines used in the study is already in use, said Dr. Rhonda Colombo, an infectious disease specialist who is the lead associate investigator for PAIVED at Madigan.
“They are readily available commercially, so if anyone went to a local pharmacy, they could receive any of the three,” Colombo said, “because they’re all FDA-approved.”
Colombo said the study is expected to wrap up enrollment by January; follow-up with participants will continue during the flu season, through next May. Study organizers will analyze results to determine comparative vaccine effectiveness in order to inform future vaccine purchasing decisions.
“The goal is to see whether one of these three is better in our military population at preventing influenza,” Colombo said, “and if we find that there’s not one that’s better, that they all appear to work equally, then that’s also very informative.”
Colombo said that type of information can help inform policy about purchasing a vaccine, because if one vaccine works better than another, Defense Department and Defense Health Agency leadership can focus on purchasing the more effective shot.
“Right now, the [Centers for Disease Control and Prevention] and Advisory Committee on Immunization Practices don’t recommend one of these vaccines over another,” Colombo said. “We want to make sure that the decision and policy is guided by science so that our beneficiaries and service members are receiving what is likely to work best."

Those volunteers taking part in the study find that it’s a little different than the usual ‘pop in, get your shot, pop out’ system of flu vaccinations.
At the enrollment desk, people find out if they’re eligible to take part in the study, provide informed consent, and give a brief medical history.
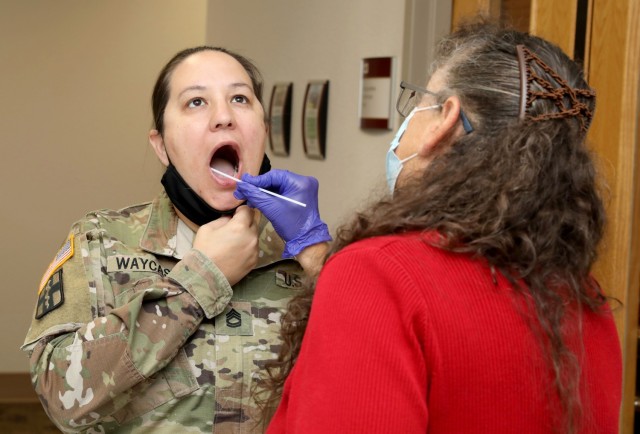
Before receiving their vaccination, participants can choose to take part in an optional cheek swab or give a blood sample as part of the study.
“We are looking at whether our genetics impact immune response and therefore make a difference in the effectiveness of the vaccine,” said Chambers.
Participants are then randomly assigned to receive one of the three FDA-approved vaccines; the type is recorded in a study database. The vaccines are randomized, meaning participants can’t pick which vaccine they’re given, and no placebos are given.
Participants are provided details on the vaccine they receive, provided a vaccine administration card and the vaccine is documented in the military immunization database and medical record.
“The vaccines are given just as they would be given at standard of care,” she said, “so they’re in the same dose and volume as you’d get routinely.”
Once participants are vaccinated, they receive small take-home kits containing a nasal swab and additional finger-stick items for them to take their own samples approximately 14 days after receiving their vaccinations.
“The reason we provide that nasal swab kit now,” Colombo said, “is that then we make sure that the participants have it available so that if they begin feeling ill, and meet certain criteria for having what we call ‘an influenza-like illness,’ they can collect their swab, which makes it more likely that we’ll be able to identify what pathogen is causing their illness.
“Hopefully, they will not become ill, and won’t ever have to use the swab,” Colombo added, “but that’s why they’re given all the instructions.”
The study is in its fourth and final year. Begun in 2018, the study was supposed to end in 2020, but with the outbreak of the COVID-19 pandemic, “The world got all turned around and upside down,” Colombo said.
“One of the interesting phenomena that occurred is that there were very low rates of influenza during 2020-2021, nationwide and within the DOD also,” she said.
“We actually had to expand the enrollment numbers and years of enrollment to include this year, so that we’re able to follow enough influenza-like illness and detect enough influenza to be able to be confident in our determination of whether or not there are any differences between the vaccines at being able to prevent influenza,” Colombo added.
Given that the world is still dealing with the COVID-19 pandemic, the study directors said that it’s important that people get vaccinated against both COVID-19 and the flu, especially since flu rates were low last year.
“With the capacity of hospitals being maxed out, it’s very important to get your flu vaccine,” said Dr. Christina Schofield, an associate professor at USUHS and principal investigator of the PAIVED study.
A recent study indicated that the possibility of a ‘twindemic’ of flu and COVID-19 this winter is quite real, and some people may be concerned about getting a COVID-19 vaccination and a flu shot close together; Schofield said the concern is unnecessary.
“You can receive those [vaccinations] at the same time. In fact, I did just that yesterday,” Schofield said, chuckling, "One in one arm, and one in the other. And the flu arm was sore first.”

Chambers said the possibility of a heavy flu season, along with the continued threat of COVID-19, reinforces the need to apply personal and public health measures to minimize the impact of circulating flu and COVID by getting vaccinated against both.
"Some military treatment facilities, such as Madigan, are administering both immunizations at the same time during mass vaccination clinics," she said.
Study organizers said the flu vaccine data collection will continue until May 2022, after which results will be tabulated in order to determine the most effective vaccine.
Studies such as the flu vaccine study give active-duty service members and other beneficiaries the opportunity to take part in world-class research, with a goal of continuing to provide top-notch medical care to America’s armed forces.
Chambers said those interested in learning more about the study can email Vivian Ramage, a registered nurse on the PAIVED study team.





Social Sharing