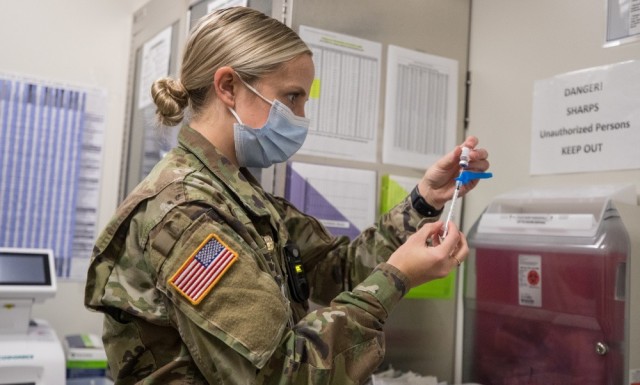
WASHINGTON -- Maj. Christine Bacsa initially hesitated to take the newly-released COVID-19 vaccine after learning she had become pregnant in December. She didn’t know if the vaccine could lead to complications with her pregnancy or if it could potentially harm her fetus.
“At that time there really wasn't enough information about the vaccine and pregnancy,” Bacsa said Thursday during a livestream panel discussing the effects of COVID-19 vaccines. “So to make my decision, I really had to think about my situation.”
However, talks with her health care team at the Office of the Army Surgeon General and other medical professionals quickly alleviated those concerns.
Bacsa, a senior Army public health nurse, successfully completed her second dose of the vaccine in April during her second trimester and experienced no side effects.
“Being pregnant, I did find myself in a similar high-risk category, given some of my own personal medical history,” Bacsa said. “I had a lot of questions about the safety of the vaccine. But really, what helped me was discussing all my concerns and questions.
“When you look at the risks and benefits … the choice in the end was really easy.”
Bacsa is now one of more than 298,600 fully-vaccinated Soldiers as of Monday. The Army also announced it had administered more than 1 million vaccines at about 80 installations worldwide and has administered 95% of the vaccines it received. Over half of active-duty Soldiers have received at least one inoculation.
“I’m incredibly proud and thankful for the effort our medical teams have invested to reach this milestone of 1 million vaccinations administered. It is a testament to their dedication to the health and safety of our force,” said Christopher Lowman, senior official performing the duties of the undersecretary of the Army. “This is helping us improve our readiness and take care of our communities.”
Although pregnant women have a slightly increased risk at developing serious illness from COVID-19, Northwestern University published data on May 11 that revealed the virus did not present any significant risk to pregnancy.
“Based on how these vaccines work in the body, it's unlikely that it'll pose a risk to us during our pregnancy or to our fetus,” Bacsa said. “These vaccines have been shown to be safe and effective.”
Bacsa said she supports vaccines as an effective treatment against coronavirus. She encourages other Soldiers to take the inoculations, as the Army strives to meet its goal of vaccinating 70% of the force by July 4.
Maj. Jessica Cassidy, chief of nursing operations at Madigan Army Medical Center at Joint Base Lewis-McChord, Washington, served at the bedside of COVID-19 patients experiencing mild respiratory problems in Texas and California in her other role as a surgical nurse with the 10th Field Hospital.
Cassidy also treated patients who required full breathing support.
“And almost all of those had bad outcomes. That is an experience that is not unique to Army medicine or Army health care providers,” said Cassidy, who has also completed her vaccinations. “That's what our health care providers across the world are dealing with. I believe that the importance of a vaccine really relies in avoiding hospitalization; avoiding that chance of needing full breathing support, because the miracle stories are so rare for those patients who make it out of there.”
Other Army medical professionals on the panel also helped dispel concerns from women considering taking the vaccine. They answered questions about the effects of vaccines on fertilization, the menstrual cycle and overall side effects.
Col. Catherine Kimball-Eayrs, a pediatrician and pediatrics consultant for the Army surgeon general, said COVID-19 vaccinations have not shown any evidence of impacting fertility or having an impact on women who are lactating.
While data has shown that women experience more side effects from taking vaccines than men, Bacsa said that could be due to more women choosing to report experiencing side effects. She added women tend to use health care services more than men and those factors combined with female physiological differences could also impact how they react to the inoculations.
Additionally, the Army analyzed data on the frequency of blood clotting disorders among patients who had taken the Johnson & Johnson vaccine. Studies found that rare cases of blood clotting tended to occur in women under age 50, but noted that there have been cases of men who also experienced clotting. After reports of such cases, the Defense Department temporarily paused distribution of the Johnson & Johnson vaccine last month before restarting again.
In two to three weeks after receiving the Johnson & Johnson vaccine, patients who suffer from severe pain, blurred vision, headaches, shortness of breath, chest or abdominal pain, bruising, or have blood spots under the skin should seek medical care immediately. Bacsa said that research has shown these to be rare cases and that the benefits of taking the J&J vaccine outweigh the risks.
Patients who take any of the vaccines may experience cold-like symptoms such as fatigue, soreness, fever and chills. However, Kimball-Eayrs said those effects typically subside within 48 hours. Bacsa said patients have concerns about the side effects should make an appointment with their health care provider or call 911, depending on the severity of the symptoms.
“We totally recognize that there absolutely are some side effects and symptoms from getting the vaccine,” Kimball-Eayrs said. “But I really do feel like the con is worth the benefit in the end.”
During the onset of the virus, Brig. Gen. Mary Krueger saw her younger Soldiers struggle to perform basic physical activity as the commander of the Tripler Army Medical Center in Honolulu. She initially had questions about the virus, as she had to care for her five children who range in age from 4 to 22.
“And then I started to see the data that came out on the level of protection that our vaccines added and that was what was really eye-opening to me,” said Krueger, now the Army Medical Command’s deputy chief of staff for support, G-1/4/6. “When somebody has gotten the disease in the community, they may or may not have a high level of antibodies, whereas after people got the vaccines, they had pretty consistent high levels of immunity.”
Kimball-Eayrs received her first vaccination dose six months ago and all of the adults in her household have been vaccinated including her husband and mother-in-law. She said that her two teenaged children have each received doses. The Centers for Disease Control recommended Thursday that children as young as 12 can take the Pfizer COVID-19 vaccination, widening the range of patients who can be inoculated against the virus.
“I'm so thrilled to be heading into the right direction and into getting out of this pandemic safely and with everybody in a healthy condition,” she said.
Cassidy said the Army medical staff have been heavily tasked in helping the harder hit civilian communities in treating infected patients. Vaccinating more patients will help Army medical care facilities return to preventative care.
“The vaccine plays a role in bringing those people back and limiting the amount of COVID-positive patients that we have in our communities to care for, so that we can bring those health care resources back to the preventative care,” Cassidy said.
The Army and Defense Department continue to partner with the Department of Health and Human Services to form the Countermeasures Acceleration Group, or CAG, a combined team designed to augment the whole-of-government approach toward combating COVID-19.
Under the CAG, formerly known as Operation Warp Speed, military logistics experts have closely managed the supply chain to help deliver more than 300 million vaccines across the country, according to CAG officials.
The U.S. government currently provides coordinated support to six vaccines in development. It has also shipped over 1 million courses of monoclonal antibody therapeutics to more than 5,800 health care facilities, officials said.
Related links:


Social Sharing