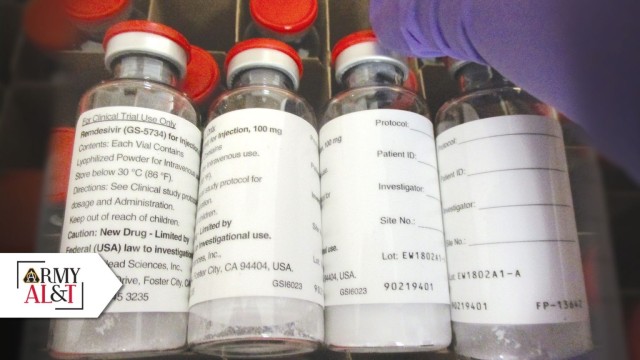
Even in extraordinary times, it’s still business as usual in the halls of the U.S. Army Medical Research and Development Command (USAMRDC). While the vast majority of Soldiers and staffers across the command have by now added a substantial dose of telework to their weekly routines in the wake of the novel coronavirus pandemic, the staffers with the Force Health Protection (FHP) team at the USAMRDC’s U.S. Army Medical Materiel Development Activity (USAMMDA) are still reporting to post daily, still fielding calls using the same office phones. Given that they stand at the very fore of the USAMRDC’s distribution of remdesivir—an investigational drug currently being provided to DOD personnel exposed to COVID-19—the daily workload takes special precedence.
“For as hard work as it’s been, it has been quite rewarding,” said Lt. Col. Sandi Parriott, FHP director. “We feel like we’re on the front lines.”
In many ways, and certainly from USAMRDC’s viewpoint, the front lines are everywhere in the fight against COVID-19. In remdesivir—a broad-spectrum antiviral treatment developed by corporate partner Gilead Sciences Inc.—there is hope that DOD soon may be able to provide a tool to help curb coronavirus infection for good. The military, based on initial data suggesting remdesivir may indeed be effective against the virus, has been studying the drug for this medical indication since early March.
How interesting then, that for all the capital, human and otherwise, invested in remdesivir, the story behind its acquisition reveals not only the importance of USAMRDC’s various scientific and legal processes, but also highlights the relationship between all the many functioning organizations surrounding—and connected to—USAMRDC.

COLLABORATIVE PARTNERS
“Working together in this space is imperative,” said Mark Hickman, a senior scientist with the Joint Program Executive Office for Chemical, Biological, Radiological and Nuclear Defense (JPEO-CBRND). “No one organization can possibly tackle the complexities of this mission alone.”
As a frequent collaborator with USAMRDC, JPEO CBRND’s Joint Project Manager for Chemical, Biological, Radiological, and Nuclear Medical (JPM CBRN Medical) is the perfect place to start when telling the story of remdesivir. Located at Fort Detrick, Maryland, JPM CBRN Medical staff enjoys a close relationship with many of USAMRDC’S internal organizations, notably USAMRDC’s U.S. Army Institute for Infectious Diseases (USAMRIID) and Walter Reed Army Institute of Research. Tasked with facilitating the advanced development and acquisition of medical countermeasures, the overall mission of the JPM CBRN Medical team is to provide the U.S. military with safe and innovative medical solutions to counter chemical, biological, radiological and nuclear threats. It is by leveraging the organization’s many relationships across private industry that brought remdesivir to the table in the first place.
According to Hickman, initial research on remdesivir began back in 2009, as it was initially developed and tested to help fight Ebola virus disease—a purpose for which, notably, it was deemed reasonably effective, according to JPM CBRN Medical leadership. Regardless, given the drug’s efficacious track record against other types of coronaviruses over the years, its application to COVID-19 was, in the words of leadership, an obvious avenue to pursue.
According to Hickman, during in vitro studies on coronaviruses isolated from bats and human lung cells, remdesivir displayed “significant” activity. Additionally, during in vivo rodent studies of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2, the virus that causes COVID-19), the early therapeutic administration of remdesivir significantly reduced lung viral load, improved clinical signs and enhanced respiratory function.
Once those facts were available, it was just a matter of picking up the phone. For JPM CBRN Medical, it was only natural to call USAMMDA.
“They happened to mention to us that [remdesivir] had some value with coronaviruses,” said Parriott, noting Force Health Protection’s “consistently productive” collaborative efforts with the team at JPM CBRN Medical, led by Lt. Col. Chuck DiTusa and Rachel Overman. “This effort with remdesivir has highlighted how well DOD organizations can work together.”
As a result, the two groups launched an effort with Gilead to create an expanded access program to provide infected Soldiers access to remdesivir (an effort aligned with Force Health Protection’s operational mission to provide investigational drugs for emergency use to the warfighter in response to a high-consequence threat). That effort—allowed under a dedicated expanded access protocol that’s been in place since March—specifically permits the investigational use of remdesivir for active-duty personnel diagnosed with moderate-to-severe novel coronavirus infection (with all doses provided by Gilead at no cost to the government). Additionally, and in this same vein, an emergency use authorization, approved by the U.S. Food and Drug Administration (FDA) in June, made remdesivir available to both military members and the greater American public. Under the authorization, Gilead donated 500,000 doses of the treatment to the federal government to be distributed, ultimately, to hospitals for the purposes of dissemination to the American public either completely free of charge or at a reduced price.
“If you have or are diagnosed with severe COVID at a civilian hospital, it is available through the EUA [emergency use authorization],” said Parriott, who additionally handles the distribution, tracking and storage of the treatment allocated to the DOD under the emergency use authorization effort.

MANY MOVING PARTS
Backing up for a second, it’s important to note that switching from one area of study to another doesn’t just happen immediately, or without substantial behind-the-scenes effort. In other words, USAMRDC efforts such as these simply can’t (and don’t) turn on a dime. In this specific case, the shift from using remdesivir as a potential Ebola medical countermeasure to its current use as a COVID-19 treatment—as well as the aforementioned expanded access protocol and emergency use authorization rulings—was the product of a process that required a unique and powerful set of legal and regulatory tools.
Indeed, for Jeremiah Kelly, who serves as chief of the FDA Regulatory Law Division, Office of the Staff Judge Advocate at USAMRDC, the process to create so-called “win-win” medical product development collaborations is similar in design to a well-choreographed dance routine.
“Each one of these collaborations with our industry partners is unique, and each one is going to require a different set of legal solutions,” said Kelly. “And even within those legal solutions, it’s almost like a radio where you have to dial into the right station—the text that governs our relationship has to reflect the equities and goals of the partners. There is no one-size-fits all model.”
DANCE OF THE CRADA
One of the tools at USAMRDC’s disposal is a type of contract known as a cooperative research and development agreement (CRADA). In short, a CRADA operates as a research-and-development tool that enables federal laboratories to transfer or contribute government resources such as personnel, services and facilities to non-federal partners—with or without reimbursement, but (as a rule) with no funding from the organization to the partner—for the purpose of conducting research or development efforts consistent with the mission of the organization. On the other side, the non-federal collaborating partner can transfer or contribute any resources to the federal organizations (including funding) toward those same research-and-development efforts.
“The moment that we heard that remdesivir had some activity against other coronaviruses, our legal team engaged with both our JPEO-CBRND and USAMMDA clients and Gilead via the CRADA mechanism to ensure that we could see the data and lay the groundwork to ensure DOD personnel would have access to this potentially effective therapy,” said Kelly, noting that the flexible nature of the CRADA enables USAMRDC to “get in the game” as early as possible. “We have so many requirements and so many capability gaps in the medical countermeasure space that the CRADA allows us to dance with whomever we want,” said Kelly. “The CRADA was the perfect legal tool to rapidly establish expanded access, with treatment courses guaranteed to DOD personnel across the globe, and with the ability of our FHP team to obtain more from Gilead as needed.”
The CRADA is further notable in that it essentially acts as a tech-transfer authority unto itself, something put into statute as part of the Stevenson-Wydler Technology Innovation Act of 1980. “It’s a very rapid non-competitive capability where we can essentially decide how we want to do it,” said Kelly. “We can provide anything to a collaborator except money, which is why it’s not competitive. And we can take in everything, including money to facilitate any collaboration. So the nice thing about the CRADA is it can almost take any shape, so long as the government’s not paying money to that collaborator.”
An example of a CRADA’s inherent flexibility is the 2016 Ebola CRADA and subsequent other-transaction agreement between Gilead and JPEO-CBRND. In that instance, the agreement included a CRADA whereby a substantial amount of the work was scheduled to take place at the USAMRDC’s USAMRIID. According to Kelly, that’s when the USAMRDC legal team stepped into the fold to ensure the various agreements were aligned and, in a turn of legal parlance, “spoke to each other.”

LEGAL ELITE
“Our legal team here at USAMRDC is actually recognized well beyond the Army in terms of its capability to provide legal solutions to facilitate FDA-regulated medical product development,” said Kelly. “Our expertise and the way that we staff these agreements is very unique to the federal government. Our attorneys are right there alongside our contract teams, making sure that the intellectual property, technical data-rights provisions and the FDA regulatory provisions are all aligned in such a way that the government’s not only going to achieve its short term goals, but it’s actually going to be structured in a way that, if the data is supporting, would result in an FDA-approved, licensed, or cleared product. We are the DOD’s recognized legal experts in medical product development, including intellectual property and FDA regulatory law. We are all passionate about our mission and bringing to bear our expertise to help our clients defeat this virus.”
According to Bob Charles, chief of medical-research law with USAMRDC, the command has anywhere between 800 to 900 active CRADAs at any given time, efforts that have led to more than $400 million in direct funding from collaborating partners for research-and-development efforts.
“I don’t think there’s any organization within the federal government that utilizes a tech transfer authority like the CRADA function as frequently as MRDC does,” said Kelly.
Opportunities for CRADAs, Charles said, normally begin at the scientist or researcher level, and can start with something as simple as a scientist reading about promising research in a journal, or perhaps even hearing about a new technology during a conference. In other words, the CRADA process employs (and encourages) a kind of grassroots, bottom-up approach to help gather initial momentum.
“Our scientists generally have a very good handle on what’s out there in the scientific community and who would be great to partner with,” said Kelly on the process. “There are folks who do a ton of market research continually to see who is doing what with what molecule out there.”
With regard to remdesivir, that kind of awareness (combined with the previously described close relationship between USAMMDA and JPM CBRN Medical) enabled all entities involved to move quickly. Kelly and his team began almost immediate development of a CRADA, and—operating through USAMMDA—secured 100 treatment courses of remdesivir (for military use only) through Gilead, with an option for an additional 100 later. For reference, 100 treatment courses is approximately 1,100 doses.
“The ability for us to rapidly use the CRADA authority to [process] remdesivir has saved lives,” said Kelly. “And that’s not hyperbole. It’s literally saved lives. It would’ve been a real question mark as to who would take the ball and run with it if we weren’t able to use our CRADA authority to rapidly partner with Gilead.” Meanwhile, he added, the other-transaction authority and CRADA mechanisms established to study remdesivir for use against Ebola continues.
LIGHTNING IN LANDSTUHL
Back in the Force Health Protection office at USAMMDA, Parriott is still manning the phones—they are, after all, available globally 24 hours a day. Despite the fact that Force Health Protection’s operational mission prohibits research (it merely reports back safety and real-world outcome data to Gilead, the FDA and other interested parties), she can’t help but marvel at the speed of the entire process. The CRADA with Gilead was signed the first week of March and evolved at a lightning-quick pace. According to Parriott, Gilead sent the first shipment of remdesivir on March 17, with FDA approval of the protocol coming down the same day. Force Health Protection enrolled the first military site in the protocol just three days later, on March 20 (at Landstuhl Regional Medical Center in Germany), with clinicians treating the first patient on March 26.
“From cooperative agreement to first patient treatment was 21 days,” said Parriott. “That’s very fast. In our immediate history, it’s definitely a record.”
Parriott further notes—in another nod to USAMMDA’s close relationship with JPM CBRN Medical—that Force Health Protection’s reputation helped drive the effort from the get-go.
“[CBRND] knew that FHP would be the quickest way to get the product out and be able to treat potential DOD members, especially [outside of the continental United States],” she said. “This shows the value of having a division like ours that can bring a product that has a lot of promise ahead of the licensure … and have value.”
The results, so far at least, have been promising. Under the expanded-access protocol, FHP had treated a total of 40 patients with remdesivir as of Aug. 25, with 36 of those (or 90 percent) now considered fully recovered after completing all protocol requirements. Of those original 40, two patients were still being actively treated under the protocol.
Still, given the entirety of these results, there can be no doubt that remdesivir holds special promise in the fight against COVID-19. So much so, in fact, that even the behind-the-scenes efforts—from the moment the antenna went up at JPM CBRN Medical to the moment USAMMDA and Gilead sat down with Jeremiah Kelly’s team to hammer out a CRADA and then beyond—are scoring (virtual) high-fives from major players themselves.
“I’m not sure anyone has a full appreciation of the tremendous work that is happening all across this command,” said Kelly. “We’re not in the business of tooting our own horn, but it’s phenomenal to see what [the command] is doing.”
Parriott can’t help but agree. So far, she points out, the information from the remdesivir clinical trial shows the treatment reduces hospitalization time and, also, the antiviral load of the patient, both of which have allowed for quicker recovery times. The FDA noted such ongoing clinical trial efforts in a statement handed down on October 22 announcing its approval of remdesivir as a treatment for COVID-19 in cases requiring hospitalization. For Parriott, watching an effort like this one—and from such a close vantage point, too—allows for a perspective that reaches far beyond the walls of her office and, to an extent, even beyond the reach of the command.
“This shows the value of having a command like ours that can bring a product that has a lot of promise ahead of the licensure and have value,” said Parriott. “We actually feel like we’re saving lives.”
For more information on the emergency-use authorization by the FDA for remdesivir, go to https://www.fda.gov/media/137564/download. To read the remdesivir fact sheet, go to https://www.fda.gov/media/137565/download. For more information on USAMRDC, go to https://mrdc.amedd.army.mil/. For more information on JPEO-CBRND, go to https://www.jpeocbrnd.osd.mil/.
RAMIN A. KHALILI is a writer for eLittle Communications Group providing support services to USAMRDC. Before assuming his current role, he spent five years as the knowledge manager for USAMRDC’s Combat Casualty Care Research Program. During his previous work as a broadcast journalist, he earned an Associated Press Award for his work in Phoenix, Arizona, before serving as Chief NASA correspondent for CBS in Orlando, Florida. He holds a B.A. in communications from Penn State University.
Subscribe to Army AL&T - the premiere source of Army acquisition news and information.




Social Sharing