An assessment by the Food and Drug Administration shows the COVID-19 vaccine developed by U.S. drug company Pfizer is around 95% effective. The vaccine will soon be considered by the FDA for emergency use authorization. If that happens, Operation Warp Speed is ready to distribute the vaccine immediately. It could happen by next week, the secretary of Health and Human Services said.
During a briefing today in Washington, Alex M. Azar said the FDA's assessment of the Pfizer vaccine shows it to be effective across different groups of Americans, including the elderly and those with comorbidities. The vaccine also did not show any significant safety concerns. He said the analysis will go before the FDA's Vaccines and Related Biological Products Advisory Committee on
Thursday. That committee will be part of determining if the vaccine can be given an emergency use authorization, or EUA.
"We could then have an EUA within days and be administering doses of vaccine to our most vulnerable next week," Azar said. "As we've discussed, based on current production schedules, we expect to have enough doses to vaccinate 20 million Americans by the end of this year, 50 million total by the end of January, and at least 100 million total by the end of the first quarter."
Army Gen. Gustave F. Perna, who serves as chief operating officer of Operation Warp Speed, said that within 24 hours of an EUA by the FDA, the vaccine could be shipped out to distribution sites across the United States to be available to the Americans who need it most.
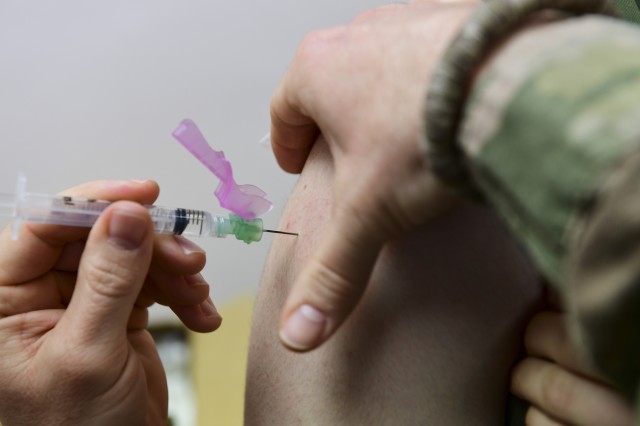

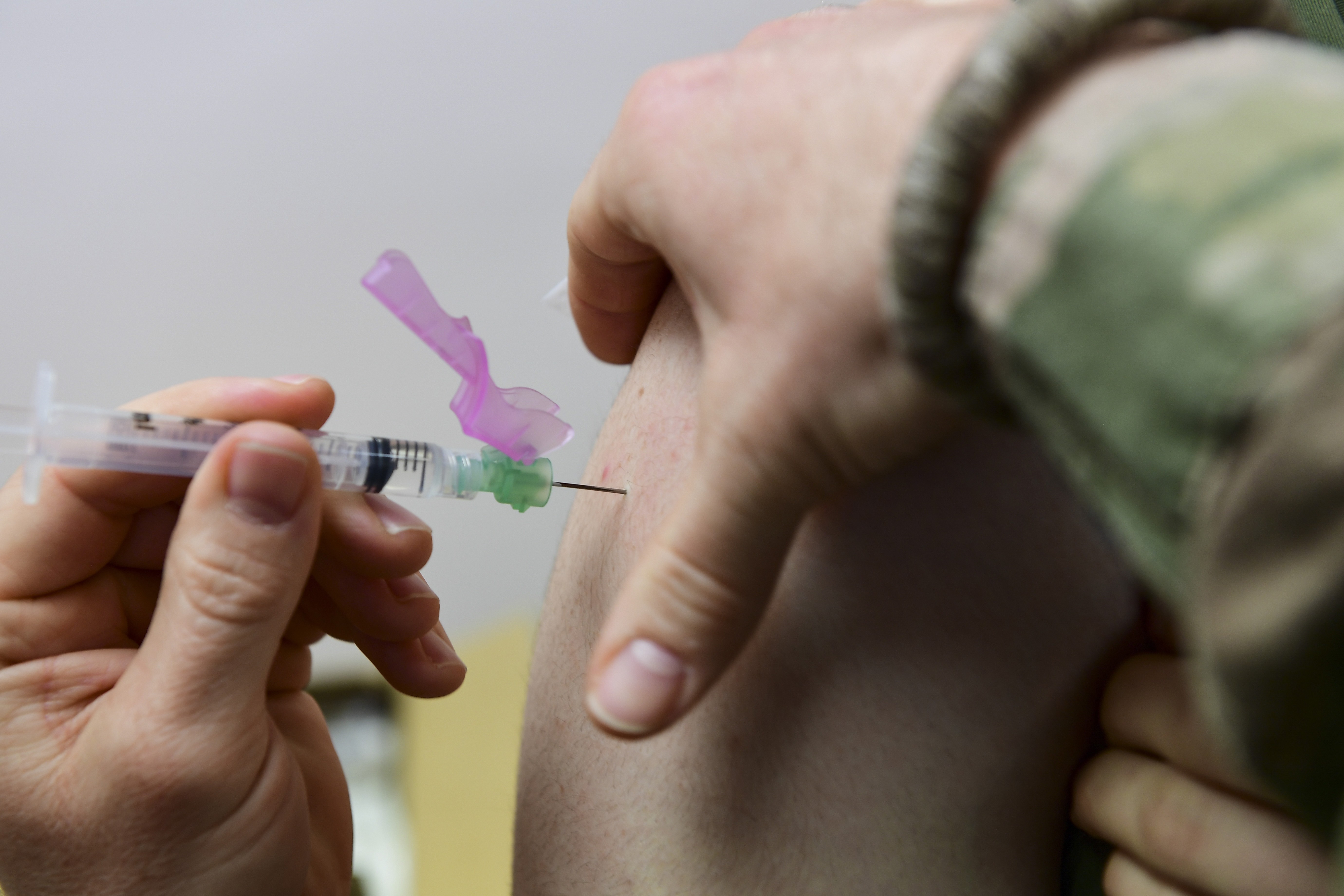
"Our goal is to make sure that we can ensure the vaccine is delivered in a timely manner, is to be delivered safely, securely, and it's ready to be utilized to start administering the vaccine as soon as possible," Perna said.
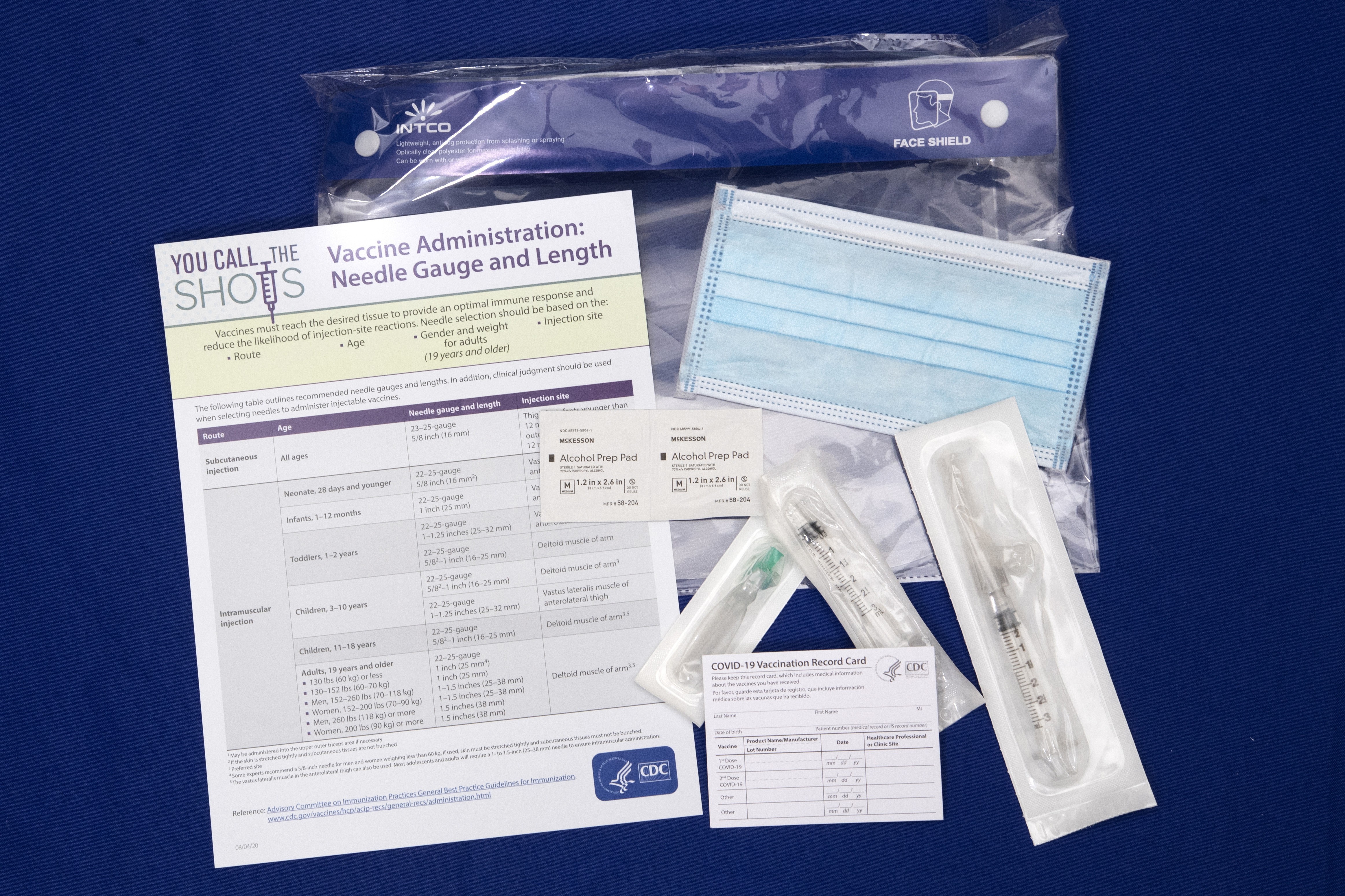
The drug alone is not enough for ensuring distribution of vaccines across the U.S., Perna said. Additional supplies are also needed to administer a drug, and the general said those supplies are also on the way to the 636 locations across 64 jurisdictions where the vaccine will be sent.
"I gave permission to already distribute syringes and needles, alcohol wipes, and diluent — which is required in support of the Pfizer vaccine," Perna said. "We'll begin that today. Distribution will be completed by Friday."
Decisions about where the vaccine goes and how much is sent to a particular location have been made in close coordination with state and jurisdiction officials.
"We are going to allocate equitably vaccine doses to all of America simultaneously," Perna said during a similar briefing in October. "As doses become available, we're pushing down and out to the United States of America."
In addition to the Pfizer vaccine, another manufacturer, Moderna, is also close to getting an EUA. Perna said OWS is ready for that as well.

"This week, we're focusing not only on the final planning for the Pfizer vaccine, but we're in deep coordination for the micro plans for the Moderna vaccine," he said. "Last night, final lock of allocations were provided to the jurisdictions. Today, tomorrow and Friday, they will lock in locations and quantities, and we'll be prepared for follow-on — eventual approval of the EUA for Moderna."
Operation Warp Speed is a partnership between the Defense Department and HHS. Specific HHS components involved include the CDC, FDA, the National Institutes of Health and the Biomedical Advanced Research and Development Authority.


Social Sharing