ABERDEEN PROVING GROUND, Md. – Although much attention in the media of late has been focused on COVID-19 vaccine candidates, a critical element in helping the Army maintain operational readiness is the ability to rapidly test large numbers of asymptomatic Soldiers in order to ensure units can safely train and deploy together. The Army Public Health Center is helping to lead this effort by supporting the Army Forces Command Combat Training Center through pooled sample testing.
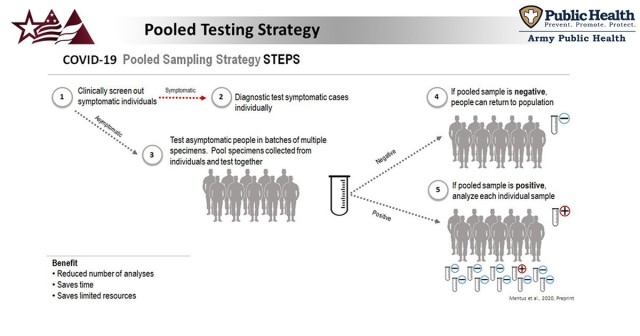
“At APHC, we conduct pooled surveillance testing, which means that we get large numbers of samples from asymptomatic individuals, and then we test those samples in groups, or “pools”, of up to 10 combined samples at once,” said Dr. Robyn Nadolny, APHC biologist and program coordinator for the COVID-19 Surveillance Testing Program. “When we see a positive result, that means a person (or multiple people) that were in that pooled sample are likely infected with the virus.”
The individuals in that pool are then sent to a clinic to have an individual test done, said Nadolny. This allows APHC to rapidly test large numbers of samples, and saves the healthcare workers at installation clinics time and reagents that are often in short supply.
“We allow the clinics to focus on testing symptomatic patients, and we focus our efforts on finding those individuals who feel just fine, but are infected with the virus and may be unknowingly spreading it to their close contacts,” said Nadolny. “This approach allows us to find those asymptomatic infections, so that those individuals can self-isolate and stop the spread of the virus.”
APHC is working with the other Army Public Health Labs around the globe to stand up a network of Public Health Labs that can support this kind of asymptomatic pooled surveillance testing. APHC began testing in July, and the lab at Public Health Command Atlantic (Fort Meade) began testing samples using similar methods in October.
“We expect the labs at Public Health Command Central and Public Health Command Pacific to be operational in the next few months,” said Nadolny. “Public Health Command Europe was the first lab to conduct this kind of testing, in the late spring of 2020.”
Polymerase chain reaction, or PCR, testing is an essential tool in the molecular biology toolbox, said Nadolny. It allows scientists to look for sections of a gene that are specific to the genetic target of interest – in this case, the SARS-CoV-2 virus that causes COVID-19.
“You start by identifying what genetic sequence you are interested in finding, and ensuring that it is unique to the organism that you want to detect,” said Nadolny. “Then, through a carefully controlled chemical reaction, you make millions of copies of this sequence of DNA or RNA, which allows you to detect the presence of the virus even at very low concentrations. This sensitivity and specificity is critical, especially when you are working with asymptomatic populations where individuals don’t know they are infected with the virus.”
The key to this approach is APHC is working with units before they deploy for their Combat Training Center rotation. The Army conducts readiness training for thousands of regular Army, Army National Guard and Army Reserve Soldiers at four Army Combat Training Centers. U.S. Army Forces Command provides mission command for two of the Combat Training Centers in the United States – the National Training Center at Fort Irwin, California, and the Joint Readiness Training Center at Fort Polk, Louisiana.
“Everyone in the unit is sampled using a nasopharyngeal swab, and those samples are sent to APHC for pooled testing several days prior to movement,” said Nadolny. “Once we find a positive pool, this is reported back to the unit point of contact, and the individuals in that pool are referred to a local clinic for individual follow-up testing to find the individual or individuals from that pool who are positive but asymptomatic.”
Once those individuals are identified, they do not move out with the rest of the unit, but are isolated until 10 days have passed since the date of their first positive test.
The APHC testing program is currently working at “initial operating capability”, which means they are largely using equipment and tools that were already in place before the pandemic struck, said Nadolny. Prior to this new mission the lab was primarily used to support the Army’s Military Tick Identification/Infection Confirmation Kit Program.
“We have been fortunate in that we have been able to execute both missions at the same time in the same space,” said Nadolny.
The Molecular Biology Section team is comprised of civilians, military personnel, and interns and fellows from the Oak Ridge Institute for Science and Education, as well as several individuals who volunteered to be temporarily detailed from other sections of APHC’s Laboratory Sciences Directorate to come and support the COVID testing mission.
“The result is a crack team of scientists who are adaptable and unflappable, and it is because of their hard work that both missions have continued to be a success during these unprecedented times,” said Nadolny.
Nadolny says the team can comfortably test around 800 samples per day in 80 pools, with the ability to stretch and test up to around 1,200 samples per day (120 pools) if needed.
In order to bring the COVID-19 testing mission online, new PCR tests needed to be identified, the components purchased, then the tests needed to be validated on the lab equipment and finally accredited by the American Association for Laboratory Accreditation, said Nadolny. New Standard Operating Procedures needed to be written for all parts of the COVID sample processing and testing program, and then all technicians needed to be trained on these new procedures.
“It wasn’t a lot of new information from a technical perspective since we were still using the same equipment and PCR was familiar to the team, but there were enough details changed that it was a lot of paperwork to get everyone officially trained and certified,” said Nadolny. “The part of the process that is the most different from the tick lab procedures is the pooling of the samples, and the purification of the samples that must be completed prior to PCR testing.”
Nadolny’s testing team is anticipating moving into a newly renovated testing facility with new equipment and an increased testing capacity.
“We are trying to reach ‘full operating capability’ at some point in the next few months, with some new high-throughput equipment – at this level we could test around 5,000 samples per day (500 pools),” said Nadolny.
Nadolny says their Army customers are happy APHC is taking an active role in COVID-19 testing.
“The more testing that can be provided, especially to asymptomatic individuals, the more we can find and prevent from spreading the infection,” said Nadolny. “We are usually able to provide results within 24 hours of receiving the samples, which is helpful to the units who need to make preparations to travel.”
The Army Public Health Center focuses on promoting healthy people, communities, animals and workplaces through the prevention of disease, injury and disability of Soldiers, military retirees, their families, veterans, Army civilian employees, and animals through population-based monitoring, investigations, and technical consultations.
Related Links:




Social Sharing