
WASHINGTON -- U.S. military personnel won't be administering any COVID-19 vaccines to the American people once the vaccines are approved for use. But the U.S. military will lend it's experienced hand in logistics to ensure the vaccine is available across the nation, said Paul Mango, the deputy chief of staff for policy at the Department of Health and Human Services.
"The overwhelming majority of Americans will get a vaccine that no federal employee, including the Department of Defense, has touched," Mango said during a Friday teleconference regarding Operation Warp Speed, the DOD and HHS effort to find a vaccine for COVID-19. "That said ... we have the best logisticians in the world at the Department of Defense, working in conjunction with the CDC, to guide ... every logistical detail you could possibly think of."
That effort, Mango said, involves things such as needles, syringes, swabs, adhesive bandages, dry ice and trucks, for instance.
"Gen. [Gustave F. Perna], and his team ... are guiding all of that with scores of folks from both the CDC and the DOD," Mango said. "We will have an operation center that will tell us at any given time exactly where every dose of vaccine is."
Those operations centers, he said, will be similar to those set up for things like hurricanes.
"We're going to have one just for vaccines that Gen. Perna, his team and the CDC are going to man 24 hours a day," Mango said. "They will know where every vaccine dose is. If a vaccine dose is at risk of expiring, they will guide the movement of that to someplace else."
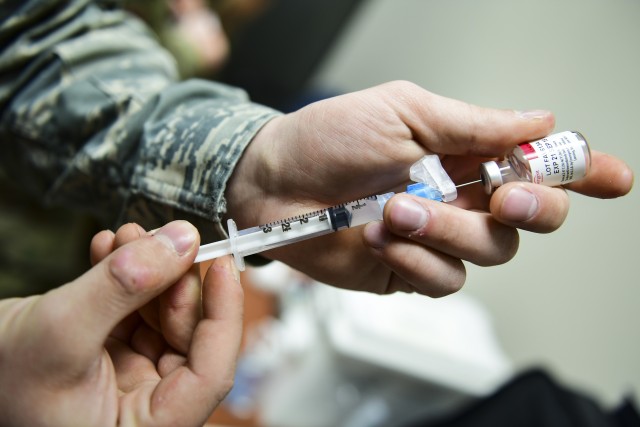
What Mango also said, however, is that federal military personnel will not be involved in touching the vaccine or administering it to Americans. He did add that if state governors want their own National Guard personnel to be involved as part of a state-run effort, they will do that at their discretion.
"The federal military will not be involved in moving any doses or injecting any vaccines," he said.

More Volunteers Needed for Vaccine Trials
Right now, there are six vaccine candidates that must be evaluated in clinical trials, and volunteers are needed to participate in those trials, said Dr. Matt Hepburn, vaccine lead for Operation Warp Speed.
"We are anticipating large-scale clinical trials — 30,000 patients each for these products," Hepburn said. "Therefore, we do need more people to be willing to sign up... if people are looking for a way that they can help us, help us as a nation, fight this pandemic, one of the ways they can do that is volunteer for these clinical trials."
While more are needed to participate in trials for vaccine candidates, many have already signed up. Dr. Jerome Adams, the U.S. surgeon general, said he's been impressed, so far, with the diversity of the candidates who've volunteered, and he said such diversity is important in a vaccine trial.
Vaccine candidate developer Moderna, for instance, announced it had completed enrollment on its Phase 3 clinical trial. Of the 30,000 participants, 37% are part of minority populations, with over 10% from African-American communities, Adams said.
"We want to applaud the recruitment outreach that's been done by Moderna," Adams said. "They were able to turn around what were initially low minority participation rates by removing barriers and meeting folks where they are and by harnessing relations with researchers who have long-standing trust with minority communities. ... We need this to continue for other trials."
Adams said those wishing to participate in trials for one of the COVID-19 vaccines can do so by visiting coronavirus.gov.
Operation Warp Speed is a partnership between the DOD and HHS. Specific HHS components involved include the CDC, the Food and Drug Administration, the National Institutes of Health and the Biomedical Advanced Research and Development Authority.
Related Links
Defense experience, capabilities further 'Herculean' Operation Warp Speed efforts
Operation Warp Speed makes swift progress
Operation Warp Speed on track for end-of-year vaccine delivery
Operation Warp Speed more than halfway to enrolling participants in vaccine trials



Social Sharing