Scientists with the U.S. Army Medical Research and Development Command (USAMRDC) confirmed during a virtual news conference today that a vaccine for COVID-19 will likely be available by the end of the year.
“It is reasonable to expect that there will be some form of a vaccine that could be available at some level to a certain population by the end of the year,” said Col. Wendy Sammons-Jackson, director of USAMRDC’s Military Infectious Diseases Research Program (MIDRP).

The news conference, which was hosted by Brig. Gen. Michael J. Talley, Commanding General of USAMRDC and Fort Detrick, Maryland, featured a slew of top USAMRDC scientists and covered a broad range of topics related to the Command’s efforts to develop a coronavirus vaccine as part its commitment to Operation Warp Speed; the aggressive public–private partnership effort initiated by the federal government last month to facilitate the development, manufacturing, and distribution of COVID-19 related countermeasures.
Currently, USAMRDC scientists are collaborating with several private sector partners on multiple vaccine candidates, one of which is likely be the first major vaccine to be tested in a Phase Three clinical trial according to statements made during the news conference. Further, scientists at USAMRDC’s Walter Reed Army Institute of Research (WRAIR) are scheduled to decide next week which of their many internally-developed vaccine candidates will move forward into the manufacturing phase, with human clinical trials of that same candidate scheduled to begin in the late summer. The data culled from these initial trials will then direct the advancement of the vaccine towards larger clinical trials near the end of the year.
“Going to a vaccine in a matter of months, from concept all the way to phase three clinical trials and potentially licensure is unprecedented, but in this case I think it’s very much possible,” said Dr. Kayvon Modjarrad, director of WRAIR’s Emerging Infectious Diseases Branch.
Said Dr. Nelson Michael, director of WRAIR’s Center for Infectious Disease Research, in an effort to provide context to the accelerated pace of coronavirus vaccine development, “Typically, getting from concept into a Phase One study is usually two-to-four years, and then getting to the time [where] you’re beginning to look at Phase Three studies can be up to ten years.”
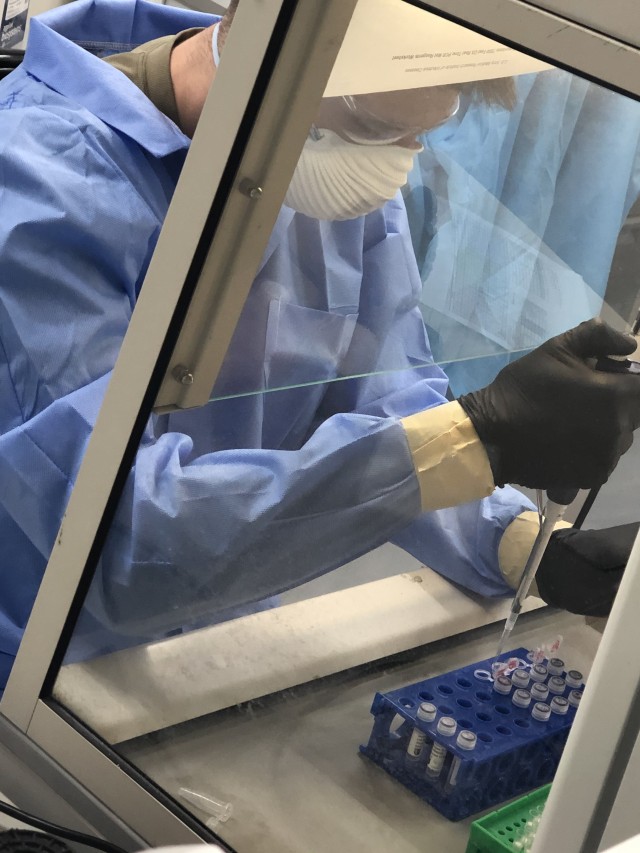
USAMRDC has been situated at the forefront of the billion-dollar research effort since the beginning of the global pandemic, with both WRAIR and USAMRDC’s U.S. Army Medical Research Institute of Infectious Diseases (USAMRIID) making significant contributions, the latter notably participating in efforts to safely replicate the virus to support countermeasure development, among other endeavors.
“As we move at top speed in the fight against COVID-19, the work being done by our scientists at both laboratories is yielding promising results,” said Talley, touting USAMRDC’s central efforts to prevent, detect, and treat the virus.
Additionally, USAMRIID is exploring possibilities in the field of therapeutics as they relate to COVID-19, moving forward on multiple lines of effort along several parallel tracks; work that includes studies on the impact of convalescent plasma, efforts to find new uses for re-purposed drugs, and, lastly, the development of monoclonal antibodies for use in various cocktails designed to stop the spread of the virus.
Elsewhere, according to Talley, USAMRDC’s U.S. Army Research Institute of Environmental Medicine (USARIEM) is evaluating the use of wearable technology to diagnose early symptoms of COVID-19, while their U.S. Army Aeromedical Research Laboratory (USAARL) is currently working to secure the airworthiness certifications required to transport COVID-19 patients requiring isolation on military aircraft.
While scientists are quick to point out that any successful vaccine would still require substantial collaboration at the federal level – allowing for policy and safety considerations, among others – they remain bullish on current timelines, as they admit their collective, singular focus on the virus in recent months, along with the requisite federal funding boost, has allowed them to learn about COVID-19 at a faster pace than any other virus before.
“This is truly an all in effort, this is something that affects the entire United States as well as the world,” said Michael.
- - -
For continuous updates on USAMRDC response to COVID-19, please visit the following website:
https://mrdc.amedd.army.mil/index.cfm/resources/covid-19
- - -
Army Futures Command
https://armyfuturescommand.com/
U.S. Army Medical Research and Development Command
USAMRDC Facebook
https://www.facebook.com/USAMRDC
USAMRDC Twitter
USAMRDC YouTube
https://www.youtube.com/user/USAMRMC
USAMRDC Flickr

Social Sharing