FORT BELVOIR, Va. (Dec. 23, 2013) -- New, bio-inspired plastics could sense and degrade chemical and biological agents, ultimately saving the lives of warfighters, first responders and civilians.
DTRA CB/JSTO-funded researchers, led by Dr. Ronald Zuckermann from Lawrence Berkeley National Laboratory, developed and characterized synthetic constructs that could lead to the creation of materials capable of protein-like sensing and catalytic degradation while providing the robustness of plastics.
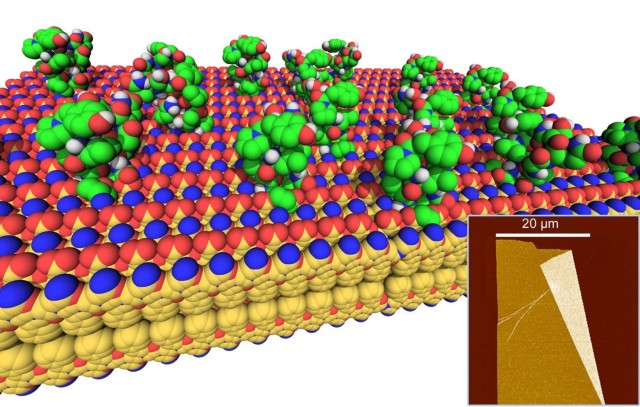
In a recently published ACS Nano article titled "Antibody-Mimetic Peptoid Nanosheets for Molecular Recognition," researchers reported the design and synthesis of a new class of protein-mimetic materials for molecular recognition: two-dimensional peptoid nanosheets decorated with functional loop domains. Peptoids are bio-inspired, sequence-specific polymers capable of folding into protein-like architectures. Because proteins are susceptible to degradation from proteases and heat and need to be confined within narrow limits of pH, there is a critical need for biomimetic materials capable of protein-like functions.
Peptoids are made from building blocks that are resistant to these hazards. Additionally, they are cheap to manufacture and offer access to a vast diversity of chemical functionalities. Earlier, this scientific team designed peptoid sequences that would self-assemble into highly stable, two-dimensional (2D) nanosheets in water (see the Journal of the American Chemical Society article titled, "Shaken, Not Stirred: Collapsing a Peptoid Monolayer To Produce Free-Floating, Stable Nanosheets"). The nanosheets were hundreds of micrometers in length and width, yet only two molecules (3 nm) thick, and could be prepared in excellent yields. This peptoid nanosheet platform provides a unique opportunity to merge the molecular recognition properties of proteins with the rugged utility of synthetic 2D nanomaterials. They are readily functionalizable on both faces, and possess high surface areas on which to display a high density of molecular recognition sites. Moreover, peptoid nanosheets are stable in air or water, do not aggregate in solution, and have a hydrophilic zwitterionic , (a molecule carrying both a positive and negative charge) exterior surface, a property that is desirable for resisting non-specific protein adsorption.
In this study, the scientists devised a strategy for displaying a wide variety of loop sequences on the nanosheet surface. They demonstrated that peptoid polymers of specific sequence can be programmed to efficiently self-assemble, or "fold," into an antibody-like architecture: a peptoid nanosheet scaffold decorated with a multiplicity of surface-exposed loop domains that are accessible for interaction with the target for binding or catalysis. The formation of loops on the surface of the nanosheet was confirmed by a variety of microscopy and scattering techniques (such as atomic force microscopy and X-ray scattering), as well as by chemical and biological function (e.g., peptide loops were accessible for molecular recognition by proteases and casein kinase II enzymes). In addition to biorecognition, they also highlight the utility of loop-displaying peptoid nanosheets as 2D templates to nucleate the "bottom-up" growth of gold nanostructures.
The creation of antibody-mimetic peptoid nanosheets demonstrates how biology's chain-folding paradigm can be extended to the world of synthetic nanomaterials. Peptoid nanosheets offer a highly designable, rugged platform for the bottom-up assembly of high surface area mesoscale 2D materials. The chemical diversity of a wide variety of molecular recognition and catalytic domains can be patterned directly into the peptoid sequence, with atomic precision, for applications ranging from nanoelectronics and chem-bio sensing, which leads to better protections of warfighters, to mineralization and catalysis.
Social Sharing